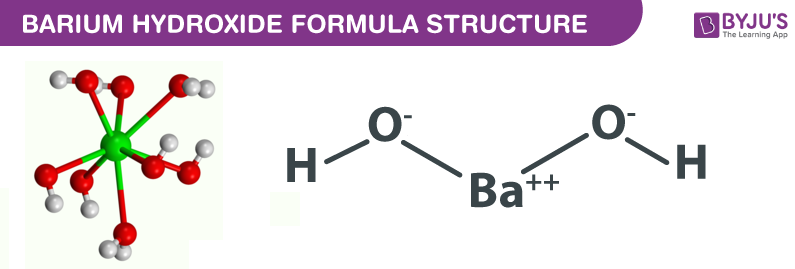
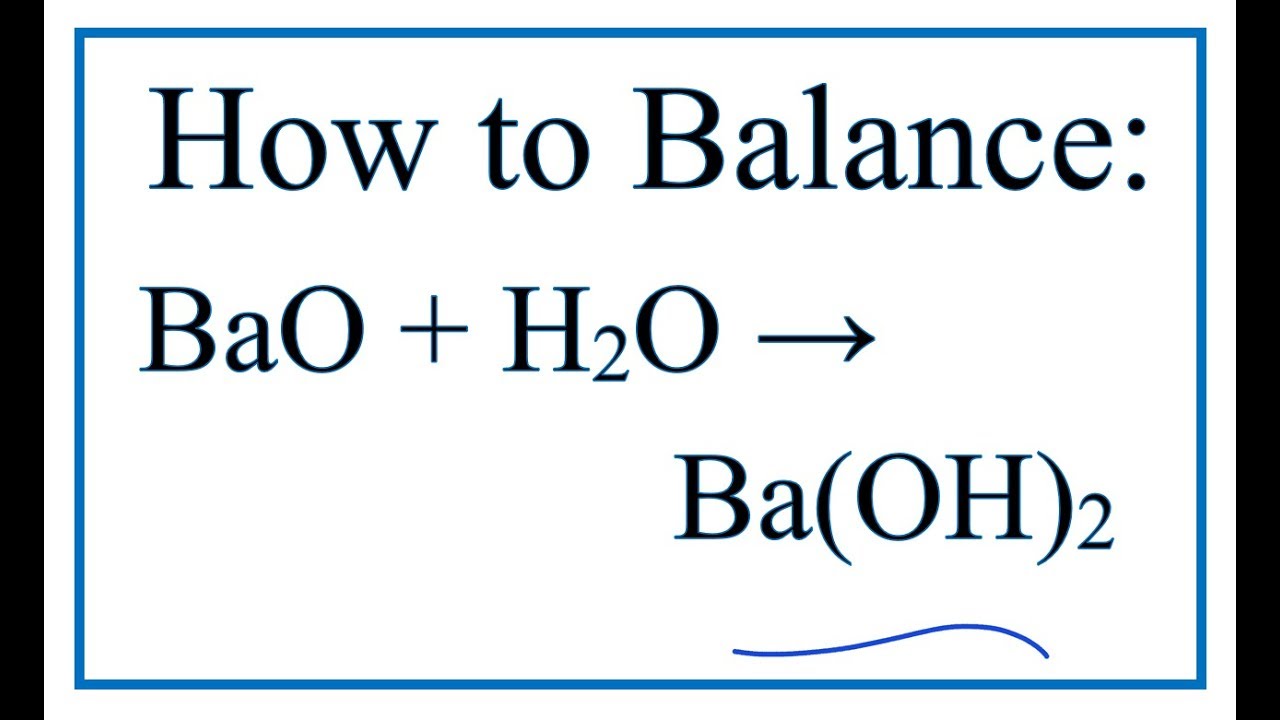
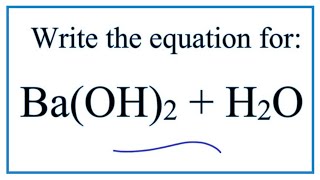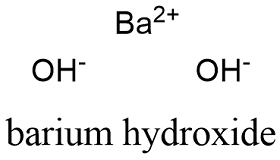Determination of chemical properties of meat: Determination of volatile basic nitrogen (VB-N), trimethylamine oxide nitrogen (TMAO-N) and trimethylamine-nitrogen (TMA-N) by Conway's micro-diffusion method (N/150 hydrochloric acid and N/70 barium hydroxide)

Question Video: Determining the Products of the Neutralization Reaction of Barium Hydroxide Ba(OH)₂ with Carbonic Acid H₂CO₃ | Nagwa

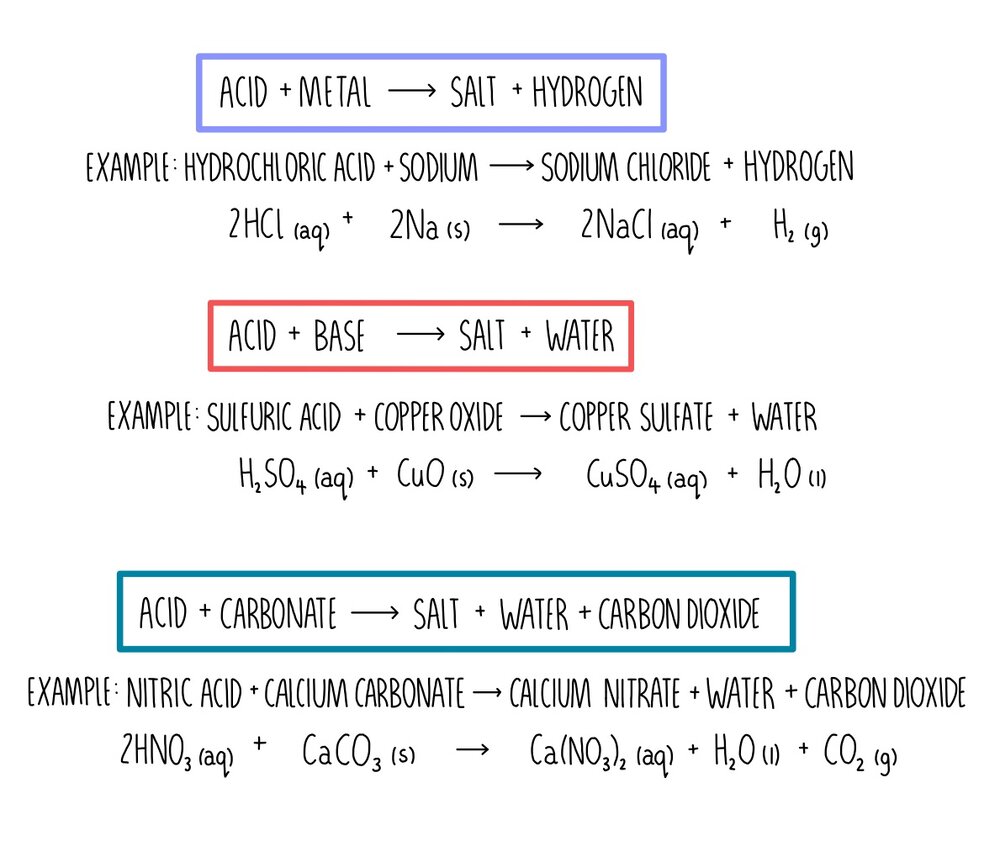
Arrange the pairs in sequence in decreasing order of basicity of an acid and acidity of a base followed by the normal salts formed by the given acid base pairs in the

Acids and Bases For now, acids ionize in aqueous solutions to form a hydrogen ion (H + ). -- “proton donors” -- monoprotic acids e.g., -- diprotic acids. - ppt download

Propylene oxide can be converted to 1,2-propanediol by the action of either dilute acid or base. When optically active propylene oxide is used, the 1,2-diol obtained from the acid hydrolysis has an

25 ml of a solution of barium hydroxide on the titration with a 0.1 molar solution of hydrochloric acid gave a titre value of 35 ml. The molarity of barium hydroxide solution is: